by Gabriel Levitt, President, PharmacyChecker.com and Prescription Justice | Nov 1, 2018 | FDA enforcement
Last week, the FDA reported on its enforcement efforts against illegal and “potentially dangerous” online drug sales in Operation Pangea XI, a global initiative run by INTERPOL in cooperation with over one hundred drug regulators. Also, President Trump signed H.R. 6, The Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act. I wish it was all about stopping rogue online pharmacies and ending the opioid crisis, but it’s not.
How Pangea XI and the SUPPORT Act are Related
The FDA is highlighting the SUPPORT Act and its effort in Pangea as important parts of the solution to stopping illegal online sales of addictive opioid drugs. The SUPPORT Act gives the FDA new authorities to stop illegal drug imports. So, what does this have to do with safe personal drug importation from pharmacies that require a prescription?
Before continuing, I can’t help note and show you that INTERPOL’s Pangea is funded by drug companies for this work. It’s this Pharma-funded initiative in which FDA plays a crucial role. The FDA does focus resources on shutting down some bad rouge sites, but it seems to also assist PhRMA in ways that curtail online access by Americans to lower-cost medicines from pharmacies in other countries.
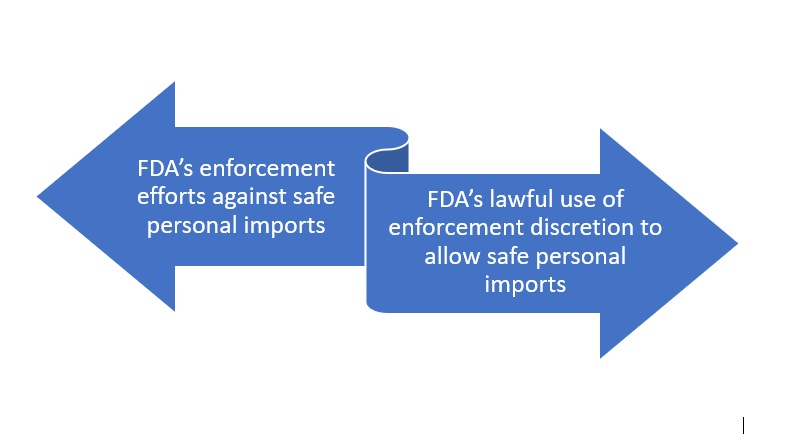
There seems to be a tug of war: FDA’s enforcement efforts against safe personal imports vs. FDA’s lawful use of enforcement discretion to allow safe personal imports.
 (more…)
(more…)
Tagged with: Enforcement, Interpol, Operation Pangea, opioid
by Gabriel Levitt, President, PharmacyChecker.com and Prescription Justice | Jun 1, 2017 | Controlled Drugs

Pharma Pushing Drugs at your Local Pharmacy
There’s a big lie peppered throughout news articles about buying medication online: 50% of medications ordered online are counterfeit, based on a report by the World Health Organization (WHO). That claim is without merit, but nonetheless pushed as fact by pharma-funded groups like Alliance for Safe Online Pharmacies. WHO never conducted such a study. In its section on counterfeit drugs, WHO used to reference an unnamed study claiming that 50% of medications ordered from illegal online pharmacies that don’t publish a mailing address are counterfeit. WHO finally removed it from its latest public education page on counterfeit drugs. That “data” has worked to scare people away from buying more affordable medication online from other countries.
What does this have to do with the opioid addiction crisis and drug company propaganda? This week it was widely reported that, essentially, drug companies fueled the epidemic of opioid abuse in America by misusing a single letter written by a doctor in 1980 about the benefits of opioids. The letter was to the New England Journal of Medicine and stated that only four people out of 40,000 who received treatment with prescription opioid medications became addicted. It was not a peer-reviewed study or even a study at all! From CBS News:
“And that is how a one-paragraph letter with no supporting information helped seed a nationwide epidemic of misuse of drugs like Vicodin and OxyContin by convincing doctors that opioids were safer than we now know them to be.”
The devil (and that’s not a euphemism) is truly in the details. The author of the letter, Dr. Hershel Jick, affirmed that the letter was meant to cover patients in a hospital setting in the short-term, not out-patient settings, long-term use, or for pick-up at a pharmacy when someone has backpain. Dr. Jick stated: “I’m essentially mortified that that letter to the editor was used as an excuse to do what these drug companies did.” (more…)
Tagged with: 1980 letter, abuse, CDC, jick, opioid, overdose, Partnership for Safe Medicine




