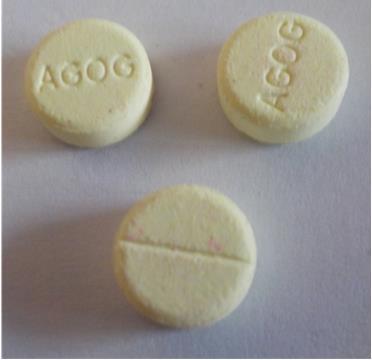
FDA Warns about “Potentially” Counterfeit Diazepam in Central Africa; We Say Buy Locally and Save!
The U.S. Food and Drug Administration (FDA) issued a warning last week that a drug product sold in Central Africa, mislabeled as diazepam, which was actually the drug haloperidol (for schizophrenia), had caused 700 adverse reactions, such as acute contractions of the muscles in the face and neck. While there are no reports that the product entered the U.S., FDA cautioned Americans who take diazepam that it could, potentially, be sold over the Internet and to be on the lookout for the pills you see in the image to your left. Sound advice!
For the backstory checkout the World Health Organization’s Medical Product Alert.
Diazepam is the generic name for the anti-anxiety medication commonly known as Valium. It is a controlled prescription drug, meaning one associated with abuse use and addiction.
We recommend to U.S. and all consumers that you not buy controlled medication internationally from an online pharmacy. Online pharmacies that sell Valium, and all controlled drugs, internationally are not eligible for the PharmacyChecker.com Verification Program. (more…)
Tagged with: diazepam, FDA warning