by Gabriel Levitt, President, PharmacyChecker.com and Prescription Justice | Jul 6, 2018 | FDA enforcement
 Last week, I published an article about new cooperation between Internet companies and the FDA undertaken to supposedly stamp out illegal opioid sales on the Internet, yet slides consciously into stopping imports of safe, lower-cost medicines. The article talked about the FDA’s Online Opioid Summit, which was held last Wednesday.
Last week, I published an article about new cooperation between Internet companies and the FDA undertaken to supposedly stamp out illegal opioid sales on the Internet, yet slides consciously into stopping imports of safe, lower-cost medicines. The article talked about the FDA’s Online Opioid Summit, which was held last Wednesday.
That summit was covered effectively (as much as possible) in this article from Tarbell to uncover further proof that the agency is, in fact, targeting online sales of regular, lower-cost medicines, which are imported for personal use. Reporter Michael McAuliff noted that the so-called public summit was mostly closed-door and very secretive. Reporters were not invited. Pharma, however, was well represented.
Apparently, it was the Internet companies who wanted privacy, perhaps to conceal efforts at stopping opioid sales on their platforms to not tip off the drug dealers. It could also be that they are ashamed of bowing to demands to censor content that Big Pharma/the FDA finds objectionable. Time will tell.
To his credit, Mr. McAuliff asks how big of a contributor to illegal opioid sales the Internet actually is. According to a division of the Centers for Disease Control and Prevention, only one tenth of one percent (0.1%) of illegally obtained prescription narcotics (opioids) are purchased online.
According to staff at the Internet Association, a trade group that represents about 40 large Internet firms, including Microsoft, Google, Amazon, Facebook, and Go Daddy, 3.4% of people abusing opioids obtain them from online sources. I have yet to find the survey, but will try to do so and write about it soon.
So how do we know that actions taken by the FDA to curtail illegal opioid sales online are being used against safe personal drug importation as well? The best example is the increased numbers of prescription orders being seized by the FDA using funding by Congress that was appropriated for stopping illegal opioid imports, particularly the drug Fentanyl and its analogs. As Tarbell finds, most such seizures are not opioids but regular prescription medicines.
The Tarbell article quotes FDA Commissioner Scott Gottlieb owning up to this:
“We’ll use [the increased funding from Congress] to interdict more illegal products flowing across our borders, including products ordered online, as well as shifting more of our criminal investigative resources to target these online sales. This is a conscious policy decision by the FDA, and we believe these online sales represent one of the highest areas of risk facing Americans right now.”
Tens of thousands of deaths occur each year from opioid overdose. A small fraction of those drugs were purchased online and those sales must be stopped! However, the tragedy of opioid abuse should not be used like this to stop people from getting more affordable medicines from Canada and other countries.
Tagged with: Enforcement, opioids
by Gabriel Levitt, President, PharmacyChecker.com and Prescription Justice | Jun 13, 2018 | FDA enforcement

For years, the pharmaceutical industry and the FDA have used the global problem of counterfeit drugs to lobby against importation of lower-cost prescription medicines. They have used the online sale of controlled drugs without a prescription as a pretext to curtail online access to licensed pharmacies in Canada. And now they’re using the opioid addiction and overdose crisis as well. So, when I learned that there will be a meeting hosted by the FDA, a “Summit” to address the problem, my radar went up. What is this really about, and who has a seat at the table?
The event is called “FDA Online Opioid Summit: Reducing Availability of Illicit Opioids Online.” I have no doubt that the participants genuinely want to stop illegal opioid sales on social media sites and rogue pharmacy sites, but Panel #1 is essentially the usual pharma suspects from the Alliance for Safe Online Pharmacies, who spent about half a million bucks lobbying Congress to stop prescription importation legislation that doesn’t even allow imports of controlled substances at all. Since 2009, that group has been pushed by Eli Lilly on the Obama administration to shut down safe international pharmacies under the guise of going after rogue sites and counterfeiters. (more…)
Tagged with: Alliance for Safe Online Pharmacies, Enforcement, opioids
by Gabriel Levitt, President, PharmacyChecker.com and Prescription Justice | Apr 19, 2018 | FDA enforcement
 As I’ve written many times, though technically illegal, patients are not prosecuted for importing medication for their own use. I like how the National Academy for State Health Policy phrases it:
As I’ve written many times, though technically illegal, patients are not prosecuted for importing medication for their own use. I like how the National Academy for State Health Policy phrases it:
“The FDA chooses to exercise enforcement discretion to not prosecute individuals who fill their prescriptions ex-U.S. so long as the drugs are for personal use and the amount does not exceed a personal-use threshold of 90 days.”
But that courtesy does not (and in many cases should not) extend to people who illegally import wholesale quantities or who import for re-sale of any kind. These people get busted. That’s precisely what happened to a New Hampshire couple, John Hayes and Plabpleung Hayes, who ended up pleading guilty to illegally importing wholesale quantities of medication and reselling it in the U.S.
This illegal drug importation threatens public health and should stand in stark contrast to filling a personal prescription from a pharmacy in Canada or other countries. Ordering medication internationally can be of great help to people who can’t afford medication here in the U.S. and should not be confused with illegal drug importation for re-sale. (more…)
Tagged with: DEA, DOJ New Hampshire, Enforcement, misbranded, wholesale importation
by Gabriel Levitt, President, PharmacyChecker.com and Prescription Justice | Mar 16, 2018 | FDA enforcement
 As reported by the Associated Press, FDA Commissioner Scott Gottlieb announced a plan earlier this month to stop imports of opioid drugs by doubling the number of inspectors at international mail facilities. This type of targeted enforcement against opioid drug products is the stuff of real public health protection, but it can easily be misused to refuse online pharmacy orders of prescription medication imports. Millions of Americans are ordering medications from abroad because they cannot afford the exorbitant prices here in the United States. Prescription medication orders from licensed pharmacies in Canada and other countries should not be conflated with illegal importation of opioids.
As reported by the Associated Press, FDA Commissioner Scott Gottlieb announced a plan earlier this month to stop imports of opioid drugs by doubling the number of inspectors at international mail facilities. This type of targeted enforcement against opioid drug products is the stuff of real public health protection, but it can easily be misused to refuse online pharmacy orders of prescription medication imports. Millions of Americans are ordering medications from abroad because they cannot afford the exorbitant prices here in the United States. Prescription medication orders from licensed pharmacies in Canada and other countries should not be conflated with illegal importation of opioids.
(more…)
Tagged with: 708, drug imports, FDA, international mail facilities, opioids, Scott Gottlieb
by Gabriel Levitt, President, PharmacyChecker.com and Prescription Justice | Dec 21, 2017 | FDA enforcement

Standing up for his constituents on drug prices
Last month, I blogged about the FDA Office of Criminal Investigations’ sending agents with search warrants to pharmacy storefronts in Florida that help older people buy more affordable medications from Canada and other countries. The FDA tried unsuccessfully to get one of the stores’ owners, Bill Hepscher, who runs Canadian MedStore, to sign a statement acknowledging that reimportation of prescription drugs is illegal. The Florida pharmacy storefront story was first reported in Kaiser Health News.
Yesterday, Senator Bill Nelson (D-FL) sent a letter to FDA Commissioner Scott Gottlieb demanding to know why some prescription orders coming from Canada and other countries were seized, and inquiring why the FDA raided the storefront offices.
Clearly, Sen. Nelson understands the technical illegality of personal drug imports and the need to protect people from counterfeit drugs and Fentanyl. However, his letter underscores his confusion about why the FDA is stopping people from getting non-controlled, regular prescription drugs now. He wants to know specifically if there is a change in FDA’s overall enforcement policy.
I’ve been warning about the serious public health ramifications of overzealous FDA enforcement on access to affordable medication for a long time. In 2015, I wrote a report about online pharmacies and personal drug importation, and sent it to the Senate Committee on Health, Education, Labor and Pensions and the House Committee on Energy and Commerce.
Back in 2006, Senator Nelson played the leading role in ending stepped-up enforcement efforts by U.S. Customs and Border Patrol (more…)
Tagged with: Bill Hepscher, Enforcement, OCI, Scott Gottlieb, Storefront
by PharmacyChecker.com | Jun 19, 2017 | FDA enforcement
On June 14 and 15, 2017, several individuals were arrested in Manitoba and British Columbia, Canada (CBC News – Manitoba) pursuant to a U.S. extradition request relating to a 2015 U.S. federal indictment of the parties on charges of illegal wholesale importation of medication into the U.S. including one batch of Avastin alleged to be counterfeit. That indictment pertains to events occurring between 2009-2011. PharmacyChecker.com strongly condemns any manufacturing and trafficking in counterfeit drugs.
Some of the individuals arrested in Manitoba, all of whom have been released on bail and are scheduled to appear in court in Canada on July 12, are also owners and/or executives of CanadaDrugs.com, Ltd. which operated a wholesale business and an online retail website. The indictment focused on the wholesale business (an area which CanadaDrugs has long since exited) and not on its website that sells medication directly to individuals for their own use pursuant to a valid prescription, does not sell Avastin, and remains licensed by the College of Pharmacists of Manitoba and an approved member of the PharmacyChecker Verification Program. We are not aware of any incident involving the sale of counterfeit medicine from CanadaDrugs.com during its 12 years operating as an approved member of our PharmacyChecker Verification Program.
We continue to closely follow this case and will post information about it, as we have done in the past.
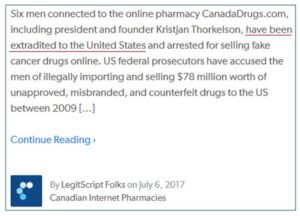
Update: It was falsely reported on the LegitScript.com blog on July 6, 2017 that individuals connected with this case “had been extradited to the United States” (as shown below). This is incorrect and no such extraditions have occurred. False and/or misleading reporting by LegitScript has occurred in the past in what appear to be attempts to foster negative perceptions of the practice of personal drug importation and of our own work in helping to make it safe. LegitScript is a company founded by John Horton, whom we have asked the House Committee on Oversight and Government Reform to investigate for possible government ethics violations.
Tagged with: canadadrugs, extradition, indictment, manitoba
 Last week, I published an article about new cooperation between Internet companies and the FDA undertaken to supposedly stamp out illegal opioid sales on the Internet, yet slides consciously into stopping imports of safe, lower-cost medicines. The article talked about the FDA’s Online Opioid Summit, which was held last Wednesday.
Last week, I published an article about new cooperation between Internet companies and the FDA undertaken to supposedly stamp out illegal opioid sales on the Internet, yet slides consciously into stopping imports of safe, lower-cost medicines. The article talked about the FDA’s Online Opioid Summit, which was held last Wednesday.






